White paper: Large-Scale LNP Process Conversions
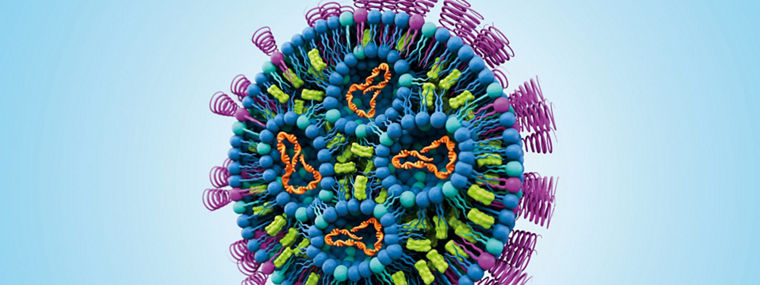
Lipid nanoparticle (LNP) formulation development begins on a small scale, often with homemade or benchtop setups, using syringe pumps and easily accessible micro-mixing systems. Once a promising formulation has been identified, the next step is to transition to standardized large-scale impingement mixing systems, such as the high flow T-mixing pumps that were used to produce the COVID-19 vaccines.
But which factors do formulators need to consider when scaling up novel LNP systems? And what hurdles can be expected?
This technical note discusses factors such as flow rates, solvent removal, risks when changing equipment and the impact of tangential flow filtration sensitivity. You can learn what needs to be considered to develop a cost- and time-efficient large-scale process.
Downloads
About our Northern Lipids® brand
Northern Lipids® is Evonik’s dedicated brand for high‑quality ionizable lipids, PEGylated lipids, and advanced formulation components used in lipid-based drug delivery. With more than 30 years of scientific heritage and Canadian roots, the brand is widely recognized in the biotech community as a trusted quality label for enabling LNPs, liposomes, and next‑generation nanomedicines.
We use the Northern Lipids® brand to identify the lipid products and technologies that support our CDMO offering - from early feasibility and process development through to GMP manufacturing and commercial supply. Whether developing vaccines, gene therapies, mRNA medicines, or other novel nucleic acid therapeutics, Northern Lipids® reflects Evonik’s commitment to delivering reliable, high‑performance lipid solutions that accelerate our customers’ innovation.
ANY QUESTIONS? JUST CONTACT US.
"Our global team of formulation experts is ready to support you with all questions around our product and service portfolio for parenteral drug delivery. Contact us to profit from more than 30 years of application know-how!"
Ahmet Alici
Business Manager